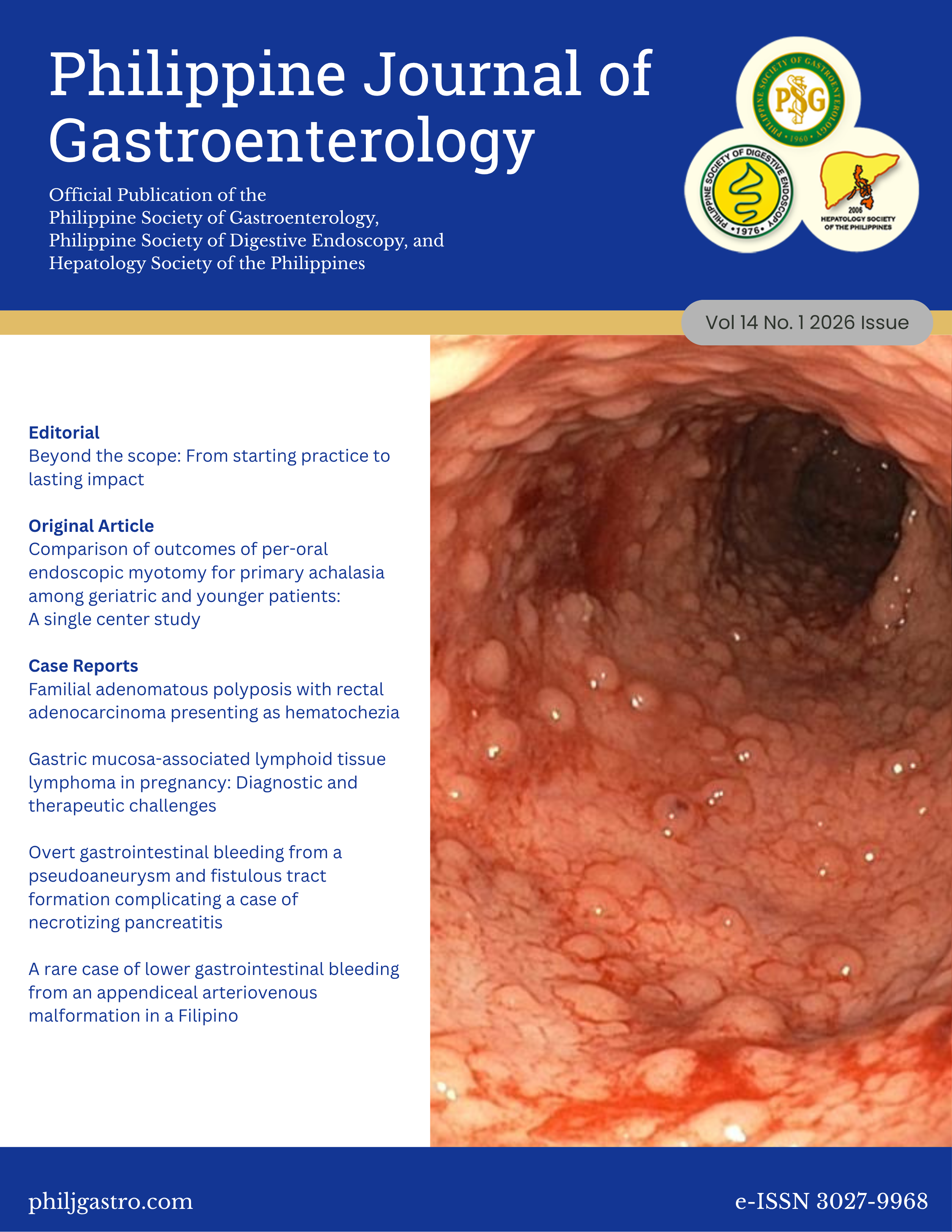
Comparison of outcomes of per-oral endoscopic myotomy for primary achalasia among geriatric and younger patients
A Single Center Study
Abstract
Background: Geriatric patients with primary achalasia have high disease burden and are often left untreated. POEM is a promising treatment for older patients with high treatment success rate and safety. Locally, there is no existing study on the comparison of POEM outcomes in young versus older patients.
Methods: Records of all primary achalasia patients who underwent POEM at our institution between January 2015 to December 2021 were reviewed. Patients were categorized into two groups: young (Sample size was determined using equivalence test. Fisher’s Exact Test or Pearson’s Chi Square Test, and Mann Whitney U Test for independent samples were employed to analyze the difference of categorical and continuous variables, respectively, between the two age groups. A p-value <0.05 was considered as statistically significant.
Results: The study included 49 patients: Group A (n=40) and Group B (n=9). The technical success rate is 100% since all patients on both groups had complete myotomy without significant differences in operative time, submucosal tunnel length, esophageal and gastric myotomy, and number of endoscopic clips. Clinical success of POEM was similar for both groups, as all patients have an Eckardt score of 0 post-POEM. Adverse event rate was low and mild in in terms of severity, with no significant difference between the two age groups. The median length of hospital stay was 5 days for both age groups.
Conclusion: This study demonstrated high clinical and technical success rate as well as low complication rate of POEM among older patients with primary achalasia similar to the results in the younger age group. With this, we recommend POEM as a safe and effective treatment option for elderly patients with primary achalasia.
Downloads
Published
License
Copyright (c) 2026 Leah Anne Legaspi, Esperanza Grace Santi, Kriska Marie Fave Atienza

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.